“The University of Pittsburgh Cancer Institute (UPCI) will be the primary site for a clinical trial of ABT-888, a drug previously proven in combination treatments to improve chemotherapy’s effectiveness by lowering cancer cells’ resistance to treatment. This trial will, for the first time, examine ABT-888 as a single agent for patients with cancers related to BRCA 1 or 2 genetic mutations, which predispose patients to breast and ovarian cancers. …”
Tag Archives: clinical trial opening
Clinical Trial Investigators Aim to Make Ovarian Cancer Cells “Terminally Ill” By Giving Advanced Ovarian Cancer Patients a Common Virus
Oncolytics Biotech Inc. (“Oncolytics”) announced today that patient enrolment has started in a Phase 1/2 clinical trial for patients with metastatic ovarian, peritoneal and fallopian tube cancers using concurrent intravenous (IV) and intraperitoneal (IP)REOLYSIN®, Oncolytics’ proprietary formulation of the human reovirus. … A cell with an activated Ras Pathway, which has lost its ability to “turn off,” leads to uncontrolled cell growth. These mutations along the Ras pathway are found in approximately two-thirds of all human cancers. The virus in REOLYSIN® will invade Ras-activated cancer cells, where the virus is able to replicate until it kills the host tumor cell. When the cancer cell dies, thousands of progeny virus particles are released, which then proceed to infect and kill adjacent cancer cells.
Oncolytics Biotech Inc. (“Oncolytics”) announced today that patient enrolment has started in a Phase 1/2 clinical trial for patients with metastatic ovarian, peritoneal and fallopian tube cancers using concurrent intravenous (IV) and intraperitoneal (IP) administration of REOLYSIN®, Oncolytics’ proprietary formulation of the human reovirus. Reovirus, an acronym for Respiratory Enteric Orphan virus, is generally believed to inhabit the respiratory and bowel systems in humans. Reovirus is found naturally in sewage and water supplies. By age 12, half of all children show evidence of reovirus exposure and by adulthood, most people have been exposed. However, the disease is non-pathogenic, meaning there are typically no symptoms from infections. The link to its cancer-killing ability was established after the reovirus was discovered to reproduce well in various cancer cell lines. Reoviruses are able to replicate only in cancer cells with an activated Ras pathway, without harming healthy cells. The Ras pathway is instrumental in transferring growth signals to the nucleus of a cell, telling the cell when and how to grow-much like an “on-off” switch.
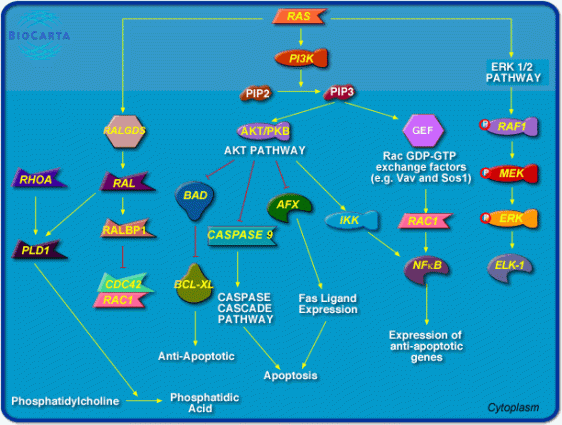 A cell with an activated Ras Pathway, which has lost its ability to “turn off,” leads to uncontrolled cell growth. These mutations along the Ras pathway are found in approximately two-thirds of all human cancers. The virus in REOLYSIN® will invade Ras-activated cancer cells, where the virus is able to replicate until it kills the host tumor cell. When the cancer cell dies, thousands of progeny virus particles are released, which then proceed to infect and kill adjacent cancer cells. The process is believed to continue until all infected cancer cells with activated Ras pathways have been infected and killed by the reovirus – all without causing the nausea, hair loss and other side effects associated with radiation and chemotherapy. More recently, Oncolytics discovered that tumor antigens generated by this virus may educate the immune system to recognize and kill tumor cells.
A cell with an activated Ras Pathway, which has lost its ability to “turn off,” leads to uncontrolled cell growth. These mutations along the Ras pathway are found in approximately two-thirds of all human cancers. The virus in REOLYSIN® will invade Ras-activated cancer cells, where the virus is able to replicate until it kills the host tumor cell. When the cancer cell dies, thousands of progeny virus particles are released, which then proceed to infect and kill adjacent cancer cells. The process is believed to continue until all infected cancer cells with activated Ras pathways have been infected and killed by the reovirus – all without causing the nausea, hair loss and other side effects associated with radiation and chemotherapy. More recently, Oncolytics discovered that tumor antigens generated by this virus may educate the immune system to recognize and kill tumor cells.
The National Cancer Institute (NCI), part of the National Institutes of Health, is sponsoring the trial under its Clinical Trials Agreement with Oncolytics, while Oncolytics will provide clinical supplies of REOLYSIN®. The Principal Investigator is Dr. David E. Cohn, Associate Professor, Division of Gynecologic Oncology at The Ohio State University College of Medicine in Columbus, Ohio.
“REOLYSIN® is an exciting agent to investigate in patients with ovarian cancer,” said Dr. Cohn. “Targeting a specific alteration commonly present in these tumors will hopefully lead to efficacy with minimal toxicity.”
“We are looking forward to working closely with the NCI to examine the effects of using REOLYSIN® with two concurrent methods of administration,” said Dr. Brad Thompson, President and CEO of Oncolytics. “Our REOLYSIN® clinical program has now expanded to include ten Phase 1/2 or Phase 2 trials in the U.S. and the U.K. using REOLYSIN® as a monotherapy or in combination with radiation or chemotherapy.”
In the Phase 1 portion of the trial, patients will receive a constant dose of IV REOLYSIN® on days 1-5 every 28 days, as well as an escalating dose of IP REOLYSIN® on days 1-2 every 28 days. In the Phase 2 portion of the study, patients will receive a constant dose of IV REOLYSIN® on days 1-5 every 28 days as well as the Maximum Tolerated Dose (MTD) of IP REOLYSIN® from the Phase 1 portion.
The primary objectives of the Phase 1 trial are to determine the safety and tolerability of IV and IP administration of REOLYSIN®, and the MTD of IP REOLYSIN® when used with a fixed dose of IV REOLYSIN®. The primary objective of the Phase 2 trial is to determine the objective response rate of treatment with IV and IP REOLYSIN® in patients with recurrent, platinum-refractory ovarian, peritoneal and fallopian tubal carcinomas. The Phase 1/2 trial is expected to enroll up to 70 patients.
[Source: Oncolytics Biotech Inc. Announces Start of Enrolment in Phase 1/2 Ovarian Cancer Clinical Trial with REOLYSIN®, Oncolytics Biotech Inc. News Release, June 10, 2008.]
Additional Information:
- The REOLYSIN® clinical trial protocol is entitled, A Phase ½ Study of Reovirus Serotype 3 – Dearing Strain (REOLYSIN®) (NSC 729968 ) in Patients With Ovarian, Primary Peritoneal and Fallopian Tube Carcinoma.
- Oncolytics Biotech Inc. Collaborators Present Positive Phase II Sarcoma Trial Results at ASCO Annual Meeting, Oncolytics Biotech Inc. News Release, June, 2, 2008.
- A Phase II Study of Intravenous REOLYSIN (Wild-type reovirus) in the Treatment of Patients with Bone and Soft Tissue Sarcomas Metastatic to the Lung, Poster Presentation, 2008 American Society of Clinical Oncology Annual Meeting (Adobe Reader PDF Document).
- A phase II study of intravenous reolysin (wild-type reovirus) in the treatment of patients with bone and soft tissue sarcomas metastatic to the lung; S. A. Soefje et. al.; J. Clin. Oncol. 26: 2008 (May 20 suppl; abstr #10568).
