“Ovarian cancer has a high case—fatality ratio, with most women not diagnosed until the disease is in its advanced stages. The United Kingdom Collaborative Trial of Ovarian Cancer Screening (UKCTOCS) is a randomised controlled trial designed to assess the effect of screening on mortality. This report summarises the outcome of the prevalence (initial) screen in UKCTOCS. …”
“Background
Ovarian cancer has a high case—fatality ratio, with most women not diagnosed until the disease is in its advanced stages. The United Kingdom Collaborative Trial of Ovarian Cancer Screening (UKCTOCS) is a randomised controlled trial designed to assess the effect of screening on mortality. This report summarises the outcome of the prevalence (initial) screen in UKCTOCS.
Methods
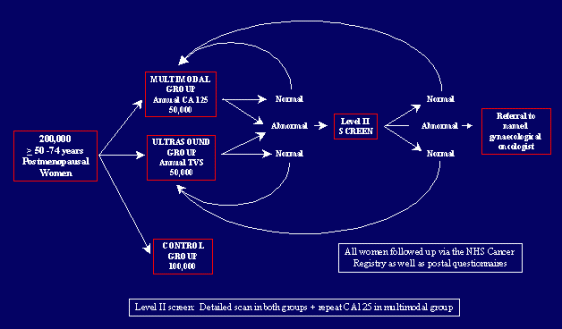
Between 2001 and 2005, a total of 202,638 post-menopausal women aged 50—74 years were randomly assigned to [1] no treatment (control; n=101,359); [2] annual CA125 screening (interpreted using a risk of ovarian cancer algorithm) with transvaginal ultrasound scan as a second-line test (multimodal screening [MMS]; n=50,640); or [3] annual screening with transvaginal ultrasound (USS; n=50,639) alone in a 2:1:1 ratio using a computer-generated random number algorithm. All women provided a blood sample at recruitment. Women randomised to the MMS group had their blood tested for CA125 and those randomised to the USS group were sent an appointment to attend for a transvaginal scan. Women with abnormal screens had repeat tests. Women with persistent abnormality on repeat screens underwent clinical evaluation and, where appropriate, surgery. This trial is registered as ISRCTN22488978 and with ClinicalTrials.gov, number NCT00058032.
Findings
In the prevalence screen, 50,078 (98.9%) women underwent MMS, and 48,230 (95.2%) underwent USS. The main reasons for withdrawal were death (two MMS, 28 USS), non-ovarian cancer or other disease (none MMS, 66 USS), removal of ovaries (five MMS, 29 USS), relocation (none MMS, 39 USS), failure to attend three appointments for the screen (72 MMS, 757 USS), and participant changing their mind (483 MMS, 1,490 USS). Overall, 4,355 of 50,078 (8.7%) women in the MMS group and 5,779 of 48,230 (12.0%) women in the USS group required a repeat test, and 167 (0.3%) women in the MMS group and 1,894 (3.9%) women in the USS group required clinical evaluation. 97 of 50,078 (0.2%) women from the MMS group and 845 of 48,230 (1.8%) from the USS group underwent surgery. 42 (MMS) and 45 (USS) primary ovarian and tubal cancers were detected, including 28 borderline tumours (eight MMS, 20 USS). 28 (16 MMS, 12 USS) of 58 (48.3%; 95% CI 35.0—61.8) of the invasive cancers were stage I/II, with no difference (p=0.396) in stage distribution between the groups. A further 13 (five MMS, eight USS) women developed primary ovarian cancer during the year after the screen. The sensitivity, specificity, and positive-predictive values for all primary ovarian and tubal cancers were 89.4%, 99.8%, and 43.3% for MMS, and 84.9%, 98.2%, and 5.3% for USS, respectively. For primary invasive epithelial ovarian and tubal cancers, the sensitivity, specificity, and positive-predictive values were 89.5%, 99.8%, and 35.1% for MMS, and 75.0%, 98.2%, and 2.8% for USS, respectively. There was a significant difference in specificity (p<0.0001) but not sensitivity between the two screening groups for both primary ovarian and tubal cancers as well as primary epithelial invasive ovarian and tubal cancers.
Interpretation
The sensitivity of the MMS and USS screening strategies is encouraging. Specificity was higher in the MMS than in the USS group, resulting in lower rates of repeat testing and surgery. This in part reflects the high prevalence of benign adnexal abnormalities and the more frequent detection of borderline tumours in the USS group. The prevalence screen has established that the screening strategies are feasible. The results of ongoing screening are awaited so that the effect of screening on mortality can be determined.
Funding
Medical Research Council, Cancer Research UK and the Department of Health, UK; with additional support from the Eve Appeal, Special Trustees of Bart’s and the London, and Special Trustees of University College London Hospital.”
Primary Source
Sensitivity and specificity of multimodal and ultrasound screening for ovarian cancer, and stage distribution of detected cancers: results of the prevalence screen of the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS); Sensitivity and specificity of multimodal and ultrasound screening for ovarian cancer, and stage distribution of detected cancers: results of the prevalence screen of the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS); Usha Menon MD, Aleksandra Gentry-Maharaj Ph.D., Rachel Hallett Ph.D. et. al, The Lancet Oncology, Early Online Publication, 11 March 2009 doi:10.1016/S1470-2045(09)70026-9.
Comment
During an interview with the New York Times, Dr. Ian Jacobs, director of the Institute for Women’s Health at University College London, and director of the trial, discussed the optimism and the caveats associated with the preliminary clinical study results as follows:
We have now demonstrated we can pick up the vast majority of women with ovarian cancer earlier than they would have otherwise been detected and before they have symptoms, .. and that a good proportion of those women have earlier stage disease than we would normally expect them to have. … [W]omen thinking of having this must understand and realize that there’s a possibility it will do more harm than good. We have reason to think it will save lives, … and then the question is, will it save enough lives to balance out the harm it does? [Emphasis added].
Robert Smith, director of cancer screening for the American Cancer Society informed the New York Times that “[w]e’re not even remotely close to knowing how to screen women of average risk with these tests, or even if we should.” Mr Smith added that it is important to run large clinical trials, but that the preliminary results of this study must be interpreted with caution.
Secondary Sources
- Screening Can Detect Early Ovarian Cancer, by Roni Caryn Rabin, Health Section, The New York Times Online Edition, March 10, 2009.
- Blood, ultrasound tests catch ovarian cancer: study, reporting by Maggie Fox, editing by Will Dunham and Paul Simao, Reuters Health News, March 10, 2009.