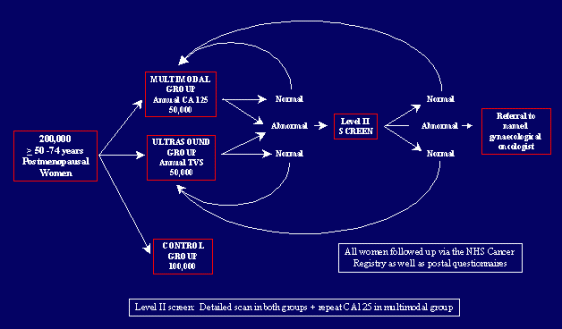
On March 10, 2009, Libby’s H*O*P*E*™ reported on the preliminary findings of a large British study that suggest that the combination of transvaginal ultrasound and CA125 blood test (a blood serum marker for ovarian cancer) can detect early ovarian cancer. A recent U.S. study, published in the April 2009 issue of Obstetrics & Gynecology, found that the same combination screening regime did not detect early stage ovarian cancer and often resulted in unnecessary surgery. The U.S. and British studies, taken together, highlight the need to find an effective screening method to detect ovarian cancer.
On March 10, 2009, Libby’s H*O*P*E*™ reported on the preliminary findings of a large British study that suggest that the combination of transvaginal ultrasound and CA125 blood test (a blood serum marker for ovarian cancer) can detect early ovarian cancer. A recent U.S. study, published in the April 2009 issue of Obstetrics & Gynecology, found that the same combination screening regime did not detect early stage ovarian cancer and often resulted in unnecessary surgery. The U.S. and British studies, taken together, highlight the need to find an effective screening method to detect ovarian cancer.

Dr. Edward E. Partridge is the Director of the University of Alabama Birmingham Comprehensive Cancer Center, Birmingham, Alabama.
In a recent interview with U.S. News & World Report, the lead researcher of the U.S. study, Dr. Edward Partridge, Director of the University of Alabama Birmingham Comprehensive Cancer Center, said, “The jury is still out on the efficacy of screening with CA125 and transvaginal ultrasound in terms of reducing the mortality rate of ovarian cancer. In this study, we do not have mortality data on the screening versus the non-screening group, so no conclusions can be made of the impact of screening with CA125 and transvaginal ultrasound.”
Partridge noted that this study only reports data on women who were screened. “We learned that the positive predictive value for the combination of tests is pretty low — in the 1 to 1.3 percent range,” he said. “A substantial number of the tests are false positives.” In addition, screening with transvaginal ultrasound lead to a higher rate of surgery for positive findings than positive CA125, Partridge said. “Transvaginal ultrasound leads to more ‘unnecessary’ surgeries,” he said. Partridge also noted that a high percentage of the cancers detected through screening were late-stage malignancies. “If you detect them at a late stage, it is unlikely that you are going to impact mortality,” he said. “In order to affect mortality, one has to detect them at an earlier stage.”
As part of the study, the U.S. researchers collected data on 34,261 women who underwent annual screening for CA125 and also had transvaginal ultrasound. A CA 125 value at or above 35 units/mL or an abnormality on transvaginal ultrasound was considered a “positive” screen. The researchers found that transvaginal ultrasound produced more positive findings for cancer than CA125 screening over the four years of screening, while the CA125 positive tests decreased from 60 percent in the first year to 34 percent in the third year. Of the 89 invasive ovarian cancers diagnosed, 60 were detected through screening. In addition, 72 percent of the screen-detected cancer were late-stage cancers, the U.S. researchers reported.
Partridge told U.S. News & World Report that even detecting cancer early may not have an impact on mortality. “In any screening trial, the ultimate test of its usefulness is does it impact mortality,” he said. Patridge added that based upon the findings of this study and The United Kingdom Collaborative Trial of Ovarian Cancer Screening (UKCTOCS) study published by Menon et. al. in the March 10 online edition of The Lancet Oncology, the CA125 blood test & ultrasound screening method will not have any effect on mortality. “What we need is a more sensitive and specific screening test,” Partridge said.
In the UKCTOCS study, a British research team found that screening was able to identify most women with gynecologic cancer. The combination of the CA125 blood test and ultrasound found 90 percent of the cancers, while ultrasound alone found 75 percent of the cancers. The researchers also found that almost 50 percent of all the cancers found were in an early stage (stage I or II). And, 48 percent of the more invasive ovarian cancers detected were designated as being stage I tumors. By way of comparison, the British researchers pointed out that only 28 percent of ovarian cancers are identified in this early stage.
Dr. David G. Mutch, the Ira C. and Judith Gall Professor of Obstetrics and Gynecology at Washington University, St. Louis, and author of an accompanying journal editorial, agreed there is no worthwhile screening test for ovarian cancer as yet. “Patients who were screened presented at the same stage as they would have if they were unscreened,” Mutch said. “There is no good screening test at this point.” Mutch added that there is no reason to screen for ovarian cancer in the general population at this point. “The prevalence of the diseases is so low, one in 2,500, and the specificity of the tests are so low, that we are going to operate on a lot of patients unnecessarily,” he said.
Primary Sources:
- Partridge E, Kreimer AR, Greenlee RT et. al. Results From Four Rounds of Ovarian Cancer Screening in a Randomized Trial. Obstet Gynecol. 2009 Apr;113(4):775-782. [PDF Document].
- Combo Screening Doesn’t Spot Early Ovarian Cancer – Even finding it early may not save lives, researchers say, by Steven Reinberg, HealthDay Article, U.S. News & World Report online, March 20, 2009.