Genentech announces positive results of Avastin Phase III study (GOG 218) in women with advanced ovarian cancer. The study showed that women who continued maintenance use of Avastin alone, after receiving Avastin in combination with chemotherapy, lived longer without the disease worsening compared to those who received chemotherapy alone. This is the first Phase III study of an anti-angiogenic therapy in advanced ovarian cancer to meet its primary endpoint.
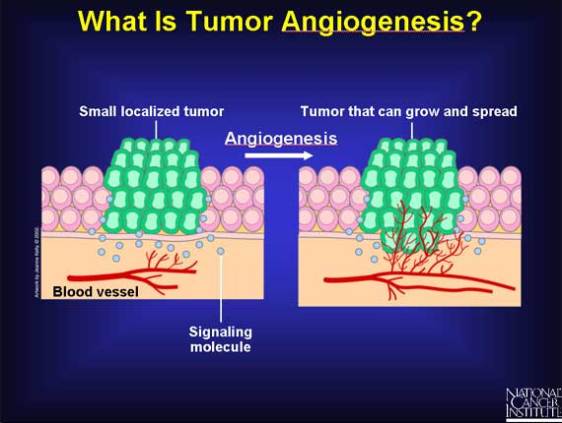
Tumor angiogenesis is the proliferation of a network of blood vessels that penetrates into cancerous growths, supplying nutrients and oxygen and removing waste products. Tumor angiogenesis actually starts with cancerous tumor cells releasing molecules that send signals to surrounding normal host tissue. This signaling activates certain genes in the host tissue that, in turn, make proteins to encourage growth of new blood vessels. Photo credit: NCI
Genentech, Inc., a wholly owned member of the Roche Group , today announced that a Phase III study showed the combination of Avastin® (bevacizumab) and chemotherapy followed by maintenance use of Avastin alone increased the time women with previously untreated advanced ovarian cancer lived without the disease worsening (progression-free survival or PFS), compared to chemotherapy alone. A preliminary assessment of safety noted adverse events previously observed in pivotal trials of Avastin. Data from the study will be submitted for presentation at the American Society of Clinical Oncology (ASCO) annual meeting, June 4 – 8, 2010.
In the three-arm study, known as Gynecologic Oncology Group (GOG) 0218, women with newly diagnosed advanced ovarian cancer who already had surgery to remove as much of the tumor as possible were randomized to receive one of the following:
- Arm 1: Placebo in combination with carboplatin and paclitaxel chemotherapy followed by placebo alone, for a total of up to 15 months of therapy
- Arm 2: Avastin in combination with carboplatin and paclitaxel chemotherapy followed by placebo alone, for a total of up to 15 months of therapy
- Arm 3: Avastin in combination with carboplatin and paclitaxel chemotherapy followed by the maintenance use of Avastin alone, for a total of up to 15 months of therapy.
The study showed that women who continued maintenance use of Avastin alone, after receiving Avastin in combination with chemotherapy (Arm 3), lived longer without the disease worsening compared to those who received chemotherapy alone. Women who received Avastin in combination with chemotherapy, but did not continue maintenance use of Avastin alone (Arm 2), did not live longer without the disease worsening compared to chemotherapy alone.
“Additional medicines are urgently needed for women with newly diagnosed advanced ovarian cancer, as most women’s cancer will worsen after their initial treatment,” said Hal Barron, M.D., F.A.C.C., Executive Vice President, Global Development and Chief Medical Officer. “We are encouraged by the positive findings of this study, which highlight the importance of continuing maintenance Avastin after combining Avastin with chemotherapy in this setting. We will discuss these results with the U.S. Food and Drug Administration.”
“This is good news for women with ovarian, primary peritoneal or fallopian tube cancers,” said GOG 0218 study chair Robert Burger, M.D., Fox-Chase Cancer Center in Philadelphia. “This study showed that after initial surgery, the combination of Avastin and chemotherapy followed by extended treatment with Avastin improves progression-free survival in women with newly diagnosed advanced tumors.”
The trial is sponsored by the National Cancer Institute (NCI) under a Cooperative Research and Development Agreement between the NCI and Genentech, and is being conducted by a network of researchers led by the GOG.
Avastin is being studied worldwide in more than 450 clinical trials for multiple types of cancer, including approximately 25 ongoing clinical trials in the United States for women with various stages of ovarian cancer.
About Ovarian Cancer
According to the American Cancer Society, ovarian cancer is the fifth leading cause of cancer death among American women. In 2009 an estimated 21,500 women were diagnosed with ovarian cancer and approximately 14,500 died from the disease in the U.S. The disease causes more deaths than any other gynecologic cancer, and the American Cancer Society estimates that nearly 70 percent of women with advanced disease will die from it within five years.
Ovarian cancer is associated with high levels of vascular endothelial growth factor (VEGF), a protein associated with tumor growth and spread. Studies have shown a correlation between a high level of VEGF and a poorer prognosis in women with ovarian cancer. Currently, treatment options for women with this disease are limited to surgery and chemotherapy.
About the GOG 0218 Study
GOG 0218 is an international, multicenter, randomized, double-blind, placebo-controlled Phase III study in 1,873 women with previously untreated advanced epithelial ovarian, primary peritoneal or fallopian tube carcinoma. The study evaluates Avastin (5 cycles) in combination with carboplatin and paclitaxel chemotherapy (6 cycles) compared to carboplatin and paclitaxel chemotherapy alone (6 cycles). The trial is also designed to assess the maintenance use of Avastin alone following the initial combined regimen of Avastin and chemotherapy (for a total of up to 15 months of therapy), compared to carboplatin and paclitaxel chemotherapy alone (6 cycles).
The primary endpoint of the study is PFS as assessed by trial investigators. Secondary and exploratory endpoints of the study include overall survival, PFS by independent review, objective response rate, safety, quality of life measures and analysis of patient tumor and blood samples.
Detailed safety assessments are ongoing. A preliminary assessment of safety performed by the GOG identified Avastin-related serious adverse events noted in previous pivotal studies, including fatal neutropenic infection and gastrointestinal perforation. The full study results, including safety information, will be presented at a future medical meeting.
About Avastin
Avastin is a solution for intravenous infusion and is a biologic antibody designed to specifically bind to a protein called VEGF. VEGF plays an important role throughout the lifecycle of the tumor to develop and maintain blood vessels, a process known as angiogenesis. Avastin interferes with the tumor blood supply by directly binding to the VEGF protein to prevent interactions with receptors on blood vessel cells. Avastin does not bind to receptors on normal or cancer cells. The tumor blood supply is thought to be critical to a tumor’s ability to grow and spread in the body (metastasize). For more information about angiogenesis, visit http://www.gene.com.
Boxed WARNINGS and Additional Important Safety Information
People treated with Avastin may experience side effects. In clinical trials, some people treated with Avastin experienced serious and sometimes fatal side effects, including:
Gastrointestinal (GI) perforation: Treatment with Avastin can result in the development of a potentially serious side effect called GI perforation, which is the development of a hole in the stomach, small intestine or large intestine. In clinical trials, this side effect occurred in more people who received Avastin than in the comparison group (0.3 percent to 2.4 percent). In some cases, GI perforation resulted in fatality.
Surgery and wound healing problems: Treatment with Avastin can lead to slow or incomplete wound healing (for example, when a surgical incision has trouble healing or staying closed). In some cases, this event resulted in fatality. Surgery and wound healing problems occurred more often in people who received Avastin than in the comparison group. Avastin therapy should not be started for at least 28 days after surgery and until the surgical wound is fully healed. The length of time between stopping Avastin and having voluntary surgery without the risk of having surgery and wound healing problems following surgery has not been determined.
Severe bleeding: Treatment with Avastin can result in serious bleeding, including coughing up blood, bleeding in the stomach, vomiting of blood, bleeding in the brain, nosebleeds and vaginal bleeding. These events occurred up to five times more often in people who received Avastin. Across cancer types, 1.2 percent to 4.6 percent of people who received Avastin experienced severe to fatal bleeding. People who have recently coughed up blood (greater than or equal to a half teaspoon of red blood) or have serious bleeding should not receive Avastin.
In clinical trials for different cancer types, there were additional serious and sometimes fatal side effects that occurred in more people who received Avastin than in those in the comparison group. The formation of an abnormal passage from parts of the body to another part (non-GI fistula formation) was seen in 0.3 percent or less of people. Severe to life-threatening stroke or heart problems were seen in 2.4 percent of people. Too much protein in the urine, which led to kidney problems, was seen in less than 1 percent of people. Additional serious side effects that occurred in more people who received Avastin than those in the comparison group included severe to life-threatening high blood pressure, which was seen in 5 percent to 18 percent of people, and nervous system and vision disturbances (reversible posterior leukoencephalopathy syndrome), which was seen in less than 0.1 percent of people. Infusion reactions with the first dose of Avastin were uncommon and occurred in less than 3 percent of people and severe reactions occurred in 0.2 percent of people.
Common side effects that occurred in more than 10 percent of people who received Avastin for different cancer types, and at least twice the rate of the comparison group, were nosebleeds, headache, high blood pressure, inflammation of the nose, too much protein in the urine, taste change, dry skin, rectal bleeding, tear production disorder, back pain and inflammation of the skin (exfoliative dermatitis). Across all trials, treatment with Avastin was permanently stopped in 8.4 percent to 21 percent of people because of side effects.
Avastin may impair fertility. Patients who are pregnant or thinking of becoming pregnant should talk with their doctor about the potential risk of loss of the pregnancy or the potential risk of Avastin to the fetus during and following Avastin therapy, and the need to continue an effective birth control method for at least six months following the last dose of Avastin.
For full Prescribing Information and Boxed WARNINGS on Avastin please visit http://www.avastin.com.
About Genentech
Founded more than 30 years ago, Genentech is a leading biotechnology company that discovers, develops, manufactures and commercializes medicines to treat patients with serious or life-threatening medical conditions. The company, a wholly owned member of the Roche Group, has headquarters in South San Francisco, California. For additional information about the company, please visit http://www.gene.com.
About The Gynecologic Oncology Group (GOG)
The Gynecologic Oncology Group is a non-profit organization of more than 300 member institutions with the purpose of promoting excellence in the quality and integrity of clinical and basic scientific research in the field of Gynecologic malignancies. The Group is committed to maintaining the highest standards in the clinical trial development, execution, analysis and distribution of results. Continuous evaluation of our processes is utilized in order to constantly improve the quality of patient care.
GOG receives support from the National Cancer Institute (NCI) of the National Institutes for Health (NIH).
Sources:
- Genentech Announces Positive Results of Avastin Phase III Study in Women with Advanced Ovarian Cancer, Press Release, Genentech, Inc., February 24, 2010.
- A Phase III Trial of Carboplatin and Paclitaxel Plus Placebo Versus Carboplatin and Paclitaxel Plus Concurrent Bevacizumab (NSC # 704865, IND #7921) Followed By Placebo, Versus Carboplatin and Paclitaxel Plus Concurrent and Extended Bevacizumab, in Women With Newly Diagnosed, Previously Untreated, Suboptimal Advanced Stage Epithelial Ovarian, Primary Peritoneal Cancer, or Fallopian Tube Cancer, Gynecologic Oncology Group ( Philip J. DiSaia ) CDR0000455114, GOG-0218, ClinicalTrials.gov (NCT00262847)[study is ongoing, but not recruiting participants].