Christina Applegate – Samantha Who?

Christina Applegate as Samantha in the ABC sitcom Samantha Who? Photo Credit: American Broadcasting Company
Recently diagnosed in July 2008 with breast cancer, Christina Applegate appeared on ABC’s Good Morning America program in August. The talented, Emmy award winning actress is currently the star of the ABC sitcom “Samantha Who?” Applegate came into the Hollywood limelight when she appeared in popular Fox sitcom “Married With Children,” in the role of “Kelly Bundy.”
With a great deal of courage, Christina revealed in the Good Morning America interview that she had a double mastecomy three weeks earlier to remove a tumor in one breast and prevent future breast cancer in the other. Christina made the decision to have a prophylatic double mastecomy because she tested positive previously for the BRCA 1 (breast cancer 1) gene mutation.
“I just wanted to kind of be rid of it,” explained Christina Applegate. “So this was the choice I made and it was a tough one.” Applegate is 36 years old, but because her mother is a two time survivor of breast cancer, Christina was carefully screened for breast cancer since she was 30 years old. “After looking at all the treatment plans, the one that was going to work for me was to have a bilateral mastectomy,” she said during the interview with ABC’s Robin Roberts, also a breast cancer survivor.
“I didn’t want to go back to the doctors every four months for testing. … I’m clear,” she declared. “Absolutely 100 percent clear and clean. It did not spread. They got everything out, so I’m definitely not going to die from breast cancer.”
To view Christina Applegate’s August 19, 2008 interview on ABC’s Good Morning America, CLICK HERE.
Jessica Queller – Pretty is What Changes

Jessica Queller, author of "Pretty Is What Changes"
Jessica Queller is a famous Hollywood writer/producer who worked on several hit television programs like Felicity, One Tree Hill, and most recently, the Gilmore Girls, which is an Emmy Award-winning, Golden Globe-nominated, American comedy-drama series. Eleven months after her mother succumbed to cancer, Jessica Queller had herself tested for the breast cancer (BRCA) gene mutation. Queller was 34 years old when she took the BRCA gene mutation blood test, and she tested positive, like Applegate, for the BRCA 1 (breast cancer) gene mutation.
Jessica’s mother had suffered from both diseases and ultimately died of ovarian cancer. In 2005, shortly after testing positive, Jessica wrote an Op-Ed piece for the New York Times entitled Cancer and the Maiden about the burden of knowledge that comes with testing positive for the breast cancer gene. This article was the launching point for her first book, a memoir, called Pretty Is What Changes: Impossible Choices, The Breast Cancer Gene and How I Defied My Destiny. Ultimately, Queller, like Applegate, decided to have both breasts removed to stave off cancer, and she wants to have her ovaries removed before she is 40 in the hope of preventing ovarian cancer in the future.
Jessica Queller is the recipient of the 2008 LIFE Hero award from the Val Skinner Foundation
To view Jessica Queller’s April 2, 2008 interview with Good Morning America’s Robbin Roberts, CLICK HERE.
What is Hereditary Breast and Ovarian Cancer?
Hereditary breast and ovarian cancer (HBOC) is identified generally by one or more of the following characteristics found in a family:
- early age onset of breast cancer (often before age 50);
- family history of both breast and ovarian cancer;
- bilateral cancers (cancer that develop in both breasts, or both ovaries, independently) or an individual with both breast and ovarian cancer;
- an autosomal dominant pattern of inheritance (vertical transmission through either the mother or father’s side of the family); and
- an increased incidence of tumors of other specific organs, such as the prostate.
Other factors that increase the chance that hereditary breast and ovarian cancer exists within a family include:
What Are BRCA 1 & BRCA 2 Genes?
In 1990, DNA linkage analysis studies on large families with the characteristics described above, identified the first gene associated with breast cancer. Scientists named this gene “breast cancer 1″ or “BRCA1.” BRCA1 mutations are transmitted in an autosomal dominant pattern within a family. Since it was clear that not all breast cancer families were linked to BRCA1, studies continued and in 1994, scientists discovered another gene similar to BRCA1, and named it “breast cancer 2″ or “BRCA2.” BRCA2 gene mutations are also transmitted in an autosomal dominant pattern within a family. If a disease is autosomal dominant, it means that an individual only needs to get the abnormal gene from one parent to inherit the disease. One of the parents may often have the disease.
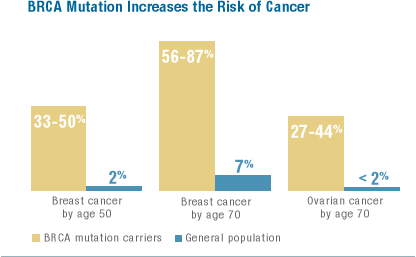
BRCA1 and BRCA2 are tumor suppressor genes, which means that they are responsible for controlling cell growth and cell death. Each individual possesses two BRCA1and two BRCA2 genes. When an individual possesses one altered or mutated copy of the BRCA1 or BRCA2 gene, the risk for various types of cancer increases:
— 36 percent to 85 percent lifetime risk for breast cancer
— 40 percent to 60 percent lifetime risk for second breast cancer (not reappearance of first tumor)
— 20 percent to 60 percent lifetime risk for ovarian cancer
— increased risk for other cancer types, such as prostate cancer
— 36 percent to 85 percent lifetime risk for breast cancer in females
— 6 percent lifetime risk for breast cancer in males
— up to 27 percent lifetime risk for ovarian cancer
— increased risk for other cancer types, such as pancreatic, prostate, laryngeal, stomach cancer, and melanoma
It is important to note that both copies of a tumor suppressor gene must be altered or mutated before a person will develop cancer. In HBOC, the first mutation is inherited from the mother or father and is therefore present in all cells of the body. This is called a “germline mutation.” Whether an individual with a germline mutation will develop cancer and where the cancer(s) will develop depends upon where (which cell type) the second mutation occurs. For example, assuming the second mutation is in the ovary, then ovarian cancer could develop. Assuming the second mutation is in the breast, breast cancer could develop. The development of a tumor ultimately requires mutations in multiple growth control genes. Loss of both copies of BRCA1 or BRCA2 is just the first step in the process. What causes these additional mutations to be acquired is unknown. Possible causes include chemical, physical, or biological environmental exposures, or cell replication errors.
An individual who possesses an inherited germline BRCA1 or BRCA2 mutation may not develop cancer in the future due to the non-occurrence of a second gene mutation which is necessary to knock out the function of the gene and start the process of tumor formation. The lack of a second gene mutation can make the cancer appear to skip generations in a family, when, in reality, one gene mutation is present. Regardless of whether cancer ultimately develops, an individual with a mutation possesses a 50/50 chance of passing the mutation on to the next generation, which could include male and/or female children. It is also important to note that the BRCA1 and BRCA2 genes are not located on the sex chromosomes, and therefore, BRCA gene mutations can be inherited from the mother’s or father’s side of the family.
What is a founder’s effect?
The majority of BRCA1 or BRCA2 gene mutations within a single family are unique. There are, however, a few exceptions. For example, specific recurring mutations have been found in individuals of Ashkenazi (Eastern Europe) Jewish descent, and persons from the Netherlands, Iceland, and Sweden. Mutations recur in these groups because of a so-called “founder’s effect.” “Founders” consist of a small group of people that interbred due to geographic or religious isolation. The “founder’s effect” occurs when that small group of people interbreeds over generations, thereby causing specific rare gene mutations to recur and become more common in the population.
The present day Ashkenazi Jewish population arose from a small group of founders. One or more of those founders probably carried specific gene mutations in BRCA1 or BRCA2. Notably, there are three mutations (two in BRCA1 and one in BRCA2) that account for the majority of the BRCA gene mutations possessed by Ashkenazi Jews. Accordingly, the existence of the founder’s effect is important to Ashkenazi Jewish individuals because it results in an increased occurrence of BRCA gene mutations in this population. This information hold practical importance within the context of gene testing, because some testing laboratories offer “ethnic-specific” gene mutation panels. Thus, laboratories can first investigate for specific gene mutations based upon the ethnic background of the indivdual, rather than search through the entire gene each time that person is tested.
In the general population, it is estimated that 1 in 500 individuals has a mutation in BRCA1 or BRCA2. In contrast, 1 in 40 Ashkenazi Jews possess recurring BRCA mutations. This increased occurrence places added emphasis on the assessment of family history for breast and ovarian cancer in Ashkenazi versus non-Ashkenazi persons.
NCI Population Estimates — Likelihood of a BRCA1 or BRCA2 Gene Mutation
The National Cancer Institute (NCI) statistics regarding the percentage of women found to possess BRCA gene mutations, as compared to samples of women and men with a variety of personal cancer histories regardless of family history, are provided below. The estimates are general in nature and cannot replace a personalized risk assessment by a certified genetic counselor, which may indicate a higher or lower mutation likelihood based upon specific family history characteristics.
Among non-Ashkenazi Jewish individuals (likelihood of having any BRCA mutation):
- General non-Ashkenazi Jewish population: 1 in 500 (.2%).
- Women with breast cancer (all ages): 1 in 50 (2%).
- Women with breast cancer (younger than 40 years): 1 in 11 (9%).
- Men with breast cancer (regardless of age): 1 in 20 (5%).
- Women with ovarian cancer (all ages): 1 in 10 (10%).
Among Ashkenazi Jewish individuals (likelihood of having one of three founder mutations):
- General Ashkenazi Jewish population: 1 in 40 (2.5%).
- Women with breast cancer (all ages): 1 in 10 (10%).
- Women with breast cancer (younger than 40 years): 1 in 3 (30%-35%).
- Men with breast cancer (regardless of age): 1 in 5 (19%).
- Women with ovarian cancer or primary peritoneal cancer (all ages): 1 in 3 (36%-41%).
Sources:
Comment: The vast majority of cancers are not due to inherited mutations. The decision to obtain genetic testing, and the action to take if you test positive for a gene mutation(s), are intensely personal decisions. It is generally recommended that you speak with a certified genetic counselor or similarly trained healthcare professional prior to engaging in genetic testing.
Additional Resources:



 Yesterday, Libby’s H*O*P*E* added a new
Yesterday, Libby’s H*O*P*E* added a new