On March 26, 2009, Senators Edward M. Kennedy (D-Massachusetts) and Kay Bailey Hutchison (R-Texas) introduced the 21st Century Cancer Access to Life-Saving Early detection, Research and Treatment (ALERT) Act, a bill to comprehensively address the challenges our nation faces in battling cancer. This is the first sweeping cancer legislation introduced since the National Cancer Act in 1971, authored by Senator Kennedy. The 21st Century Cancer ALERT Act is a comprehensive approach to cancer prevention and detection, research and treatment. It invests in cancer research infrastructure and improves collaboration among existing efforts. Prevention and early detection for those most at risk are emphasized through support for innovative initiatives and new technologies such as biomarkers. The legislation addresses the need to increase enrollment in clinical research by increasing access and removing barriers to patients’ participation in clinical trials. The bill also includes a plan designed to improve care for cancer survivors. Additional provisions regarding prevention and screening initiatives will increase access to care for underserved populations and reduce the burden of disease and cost of healthcare to the nation.

Edward M. Kennedy, U.S. Senator For The Commonwealth of Massachusetts
On March 26, 2009, Senators Edward M. Kennedy (D-Massachusetts) and Kay Bailey Hutchison (R-Texas) introduced the 21st Century Cancer Access to Life-Saving Early detection, Research and Treatment (ALERT) Act, a bill to comprehensively address the challenges our nation faces in battling cancer. This is the first sweeping cancer legislation introduced since the National Cancer Act in 1971, authored by Senator Kennedy. The 21st Century Cancer ALERT Act is a comprehensive approach to cancer prevention and detection, research and treatment. It invests in cancer research infrastructure and improves collaboration among existing efforts. Prevention and early detection for those most at risk are emphasized through support for innovative initiatives and new technologies such as biomarkers. The legislation addresses the need to increase enrollment in clinical research by increasing access and removing barriers to patients’ participation in clinical trials. The bill also includes a plan designed to improve care for cancer survivors. Additional provisions regarding prevention and screening initiatives will increase access to care for underserved populations and reduce the burden of disease and cost of healthcare to the nation.
We provide below the full text of the following documents:
_____________________________________________________
KENNEDY ON THE INTRODUCTION OF THE 21st Century ALERT Act
As Entered into the [Congressional] Record
March 26, 2009
FOR IMMEDIATE RELEASE
Thirty seven years ago, a Republican President and Democratic Congress came together in a new commitment to find a cure for cancer. At the time, a cancer diagnosis meant almost certain death. In 1971, we took action against this deadly disease and passed the National Cancer Act with broad bipartisan support, and it marked the beginning of the War on Cancer.
Since then, significant progress has been made. Amazing scientific research has led to methods to prevent cancer, and treatments that give us more beneficial and humane ways to deal with the illness. The discoveries of basic research, the use of large scale clinical trials, the development of new drugs, and the special focus on prevention and early detection have led to breakthroughs unimaginable only a generation ago.
As a result, cancer today is no longer the automatic death sentence that it was when the war began. But despite the advances we have made against cancer, other changes such as aging of the population, emerging environmental issues, and unhealthy behavior, have allowed cancer to persist. The lives of vast numbers of Americans have been touched by the disease. In 2008, over 1.4 million Americans were diagnosed with some form of cancer, and more than half a million lost their lives to the disease.
The solution isn’t easy but there are steps we can and must take now, if we hope to see the diagnosis rate decline substantially and the survival rate increase in the years ahead. The immediate challenge we face is to reduce the barriers that obstruct progress in cancer research and treatment by integrating our current fragmented and piecemeal system of addressing the disease.
Last year, my colleague Senator Hutchison and I agreed that to build on what the nation has accomplished, we must launch a new and more urgent war on cancer. The 21st Century Cancer ALERT Act we are introducing today will accelerate our progress by using a better approach to fighting this relentless disease. Our goal is to break down the many barriers that impede cancer research and prevent patients from obtaining the treatment that can save their lives.
We must do more to prevent cancer, by emphasizing scientifically proven methods such as tobacco cessation, healthy eating, and exercise. Healthy families and communities that have access to nutritious foods and high quality preventive health care will be our best defense against the disease. I’m confident that swift action on national health reform will make our vision of a healthier nation a reality. Obviously, we cannot prevent all cancers, so it is also essential that the cancers that do arise be diagnosed at an initial, curable stage, with all Americans receiving the best possible care to achieve that goal.
We cannot overemphasize the value of the rigorous scientific efforts that have produced the progress we have made so far. To enhance these efforts, our bill invests in two key aspects of cancer research– infrastructure and collaboration of the researchers. We include programs that will bring resources to the types of cancer we least understand. We invest in scientists who are committed to translating basic research into clinical practice, so that new knowledge will be brought to the patients who will most benefit from it.
One of the most promising new breakthroughs is in identifying and monitoring the biomarkers that leave enough evidence in the body to alert clinicians to subtle signs that cancer may be developing. Biomarkers are the new frontier for improving the lives of cancer patients because they can lead to the earliest possible detection of cancer, and the Cancer ALERT Act will support the development of this revolutionary biomarker technology.
In addition, we give new focus to clinical trials, which have been the cornerstones of our progress in treating cancer in recent decades. Only through clinical trials are we able to discover which treatments truly work. Today, however, less than 5% of cancer patients currently are enrolled in clinical trials, because of the many barriers exist that prevent both providers and patients from participating in these trials. A primary goal of our bill is to begin removing these barriers and expanding access to clinical trials for many more patients.
Further, since many cancer survivors are now living longer lives, our health systems must be able to accommodate these men and women who are successfully fighting against this deadly disease. It’s imperative for health professionals to have the support they need to care for these survivors. To bring good lifelong care to cancer survivors, we must invest more in research to understand the later effects of cancer and how treatments affect survivors’ health and the quality of their lives.
We stand today on the threshold of unprecedented new advances in this era of extraordinary discoveries in the life sciences, especially in personalized medicine, early diagnosis of cancer at the molecular level, and astonishing new treatments based on a patient’s own DNA. To make the remarkable promise of this new era a reality, we must make sure that patients can take DNA tests, free of the fear that their genetic information will somehow be used to discriminate against them. We took a major step toward unlocking the potential of this new era by approving strong protections against genetic discrimination in health insurance and employment when the Genetic Nondiscrimination Act was signed into law last year.
In sum, we need a new model for research, prevention and treatment of cancer, and we are here today to start that debate in Congress. We must move from a magic bullet approach to a broad mosaic of care, in which survivorship is also a key part of our approach to cancer. By doing so, we can take a giant step toward reducing or even eliminating the burden of cancer in our nation and the world. It’s no longer an impossible dream, but a real possibility for the future.
###
Press Contact
Anthony Coley/ Melissa Wagoner (202) 224-2633
___________________________________________
Kennedy Renews the War Against Cancer
March 26, 2009
FOR IMMEDIATE RELEASE
Bill will Renew America’s Commitment to Fighting Cancer and Finding Cures
WASHINGTON, DC— Senators Edward M. Kennedy and Kay Bailey Hutchison today introduced the 21st Century Cancer Access to Life-Saving Early detection, Research and Treatment (ALERT) Act, a bill to comprehensively address the challenges our nation faces in battling this disease. This is the first sweeping cancer legislation introduced since the National Cancer Act in 1971, authored by Kennedy.
The 21st Century Cancer ALERT Act will provide critical funding for promising research in early detection, and supply grants for screening and referrals for treatment. These measures will also ensure patient access to prevention and early detection, which is supplemented by increased access to clinical trials and information.
The bill places an emphasis on strengthening cancer research and the urgent need for resources to both prevent and detect cancers at an early stage. The bill strives to give scientists the tools they need to fight cancer and to understand more thoroughly how the disease works. Through fostering new treatments, increased preventative measures and funding for research, the ALERT Act begins a new chapter in how Americans will live with and fight cancer.
Senators Kennedy and Hutchison first proposed the idea for comprehensive cancer legislation last May, when the Health, Education, Labor and Pensions Committee held a hearing to discuss the need for a renewed focus on the deadly disease. Elizabeth Edwards, Lance Armstrong and Hala Moddelmog from Susan G. Komen for the Cure testified at the hearing.
Senator Kennedy, Chairman of the Health, Education, Labor, and Pensions Committee, said, “We’ve come a long way in fighting cancer since we passed the National Cancer Act thirty-eight years ago, but not far enough. Americans still live in fear that they or someone they love will be affected. Today, we’re better equipped for the fight— learning each and every day a little bit more about the disease and what we can do to fight it. Cancer is a complex disease and it requires comprehensive strategies to fight it— strategies that integrate research, prevention and treatment. This bill will renew our efforts to make progress in the battle against cancer, and to give patients and their families a renewed sense of hope.”
“Our nation declared the War on Cancer in 1971, yet, nearly 38 years later, cancer is expected to become the leading killer of Americans. We must bring renewed focus and vigor to this fight.” said Senator Hutchison. “The prescription isn’t simple, but there are steps we must take if we are going to see the cancer diagnosis rate decline, while raising the prognosis for survival among those who do have the disease. Our legislation will enact those necessary steps so we may see more progress and coordination in cancer research and treatment.”
“We know how to lengthen and improve the lives of people with cancer, but we’ve chosen as a nation to turn our backs on some of us who have the disease,” said Elizabeth Edwards. “I urge the United States Senate to embrace the ALERT Act and get it to the President’s desk as soon as possible.”
“In 2010, cancer is expected to be the leading cause of death worldwide. Every American is touched by this disease,” said Lance Armstrong, chairman and founder of the Lance Armstrong Foundation. “The 21st Century Cancer ALERT Act and its authors’ leadership in reforming our nation’s approach to the war on cancer are a very welcome step forward to every member of the LIVESTRONG movement.”
“It’s been 38 years since our nation first declared war on cancer, and yet we are still facing a significant cancer crisis. The Kennedy-Hutchison Cancer ALERT Act will reignite the war on cancer,” said Nancy G. Brinker, founder of Susan G. Komen for the Cure. “We must all work together and let nothing stand in the way of discovering and delivering the cures to cancer.”
Senate action on this bill is expected this Congressional session.
A section-by-section summary of the legislation is below as well as an op-ed authored by Senators Hutchison and Kennedy that appeared this morning in the Houston Chronicle and on the Boston Globe’s website.
_____________________________________________
21st Century Cancer ALERT Act
Senators Kennedy and Hutchison
Section by Section Summary
The 21st Century Cancer ALERT Act is a comprehensive approach to cancer prevention and detection, research and treatment. It invests in cancer research infrastructure and improves collaboration among existing efforts. Prevention and early detection for those most at risk are emphasized through support for innovative initiatives and new technologies such as biomarkers. The legislation addresses the need to increase enrollment in clinical research by increasing access and removing barriers to patients’ participation in clinical trials. The bill also includes a plan designed to improve care for cancer survivors. Additional provisions regarding prevention and screening initiatives will increase access to care for underserved populations and reduce the burden of disease and cost of healthcare to the nation.
Section 1 and 2 – Findings and Declaration of Purpose
Section 3- Advancement of the National Cancer Program (NCP)
Modernize the role of the National Cancer Institute (NCI) in coordinating the NCP
- Identifies relevant federal agencies to coordinate with NCI
- Improves the annual budget estimate for the NCP by including the needs of the entire NCP and submitting the budget annually to House and Senate Budget and Appropriations Committees
- Increases participation of other federal agencies in the National Cancer Advisory Board
- Encourages early detection and translational research opportunities
Biological Resource Coordination and Advancement of Technologies for Cancer Research
Section 4 – Comprehensive and Responsible Access to Research, Data, and Outcomes
- Calls for guidance from the Office of Human Research Protection on the use of a centralized Institutional Review Board
- Improves privacy standards in clinical research by clarifying when de-identified patient information may be disclosed
- Calls for HHS to study the advantages and disadvantages of the synchronization of the standards for research under the Common Rule and the Privacy Rule
- Clarifies the application of the Privacy Rule to external researchers
Section 5- Enhanced Focus and Reporting on Cancer Research
- Calls for NCI to report annually on plans and progress regarding research on cancers with low incidence and low survival rates
- Establishes grants program to conduct research on cancers with low incidence and low survival rates
Section 6 – Continuing Access to Care for Prevention and Early Detection
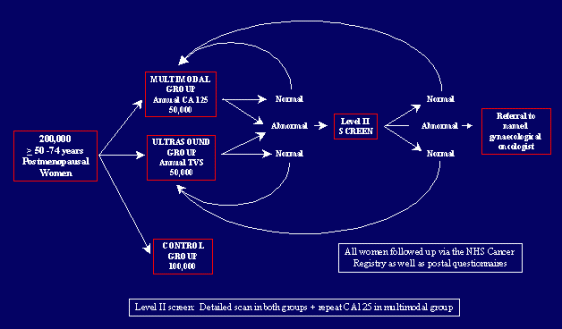
Screening and Early Detection
Cancer Prevention
- Authorizes grants for a medical mobile van program to conduct cancer screening and prevention education activities in communities that are underserved and suffer from barriers to preventative cancer care
Access to Prevention and Early Detection for Certain Cancers
Section 7– Early Recognition and Treatment of Cancer Through the Use of Biomarkers
Promote the Discovery and Development of Biomarkers
- Establishes and coordinates federal agencies to establish a highly directed, contract based program that will support the development of innovative biomarker discovery technologies
- Calls for FDA and CMS to work together to create guidelines for clinical study designs that will enable sponsors to generate clinical data that will be adequate for review by both agencies
- Conducts a demonstration project to provide limited regional coverage for biomarker tests and establish procedures for independent research entities to conduct high quality assessments of the efficacy and cost effectiveness of biomarker tests
Section 8: National Cancer Coverage Guidelines
Ensure Patient Access to Clinical Trials
- Facilitates expanded access to clinical trials by requiring ERISA governed health plans to continue to provide coverage of routine care regardless of whether a patient enrolls in a clinical trial
Section 9: Health Professions Workforce
Ensure a Stable Workforce for the Future
Section 10: Patient Navigator Program
Improve Upon Existing Patient Navigator Programs
- Ensures that patient navigators meet minimum core proficiencies
- Reauthorizes the Patient Navigator program through 2015
Section 11: Cancer Care and Coverage Under Medicaid and Medicare
Improvements in Coverage of Cancer Services
- Codifies current Medicare policy to reimburse for routine care while patients are enrolled in clinical trials
- Conducts a demonstration project to evaluate the cost, effectiveness, and potential savings to Medicare of reimbursing providers for comprehensive cancer care planning services to the Medicare population
- Directs states to offer tobacco cessation medications and counseling to pregnant women enrolled in Medicaid
Section 12: Cancer Survivorship and Complete Recovery Initiatives
Childhood Cancers
- Establishes priority areas for NIH activities related to childhood cancer survivorship
- Authorizes grants for research on the causes of health disparities in childhood cancer survivorship and to evaluate follow up care for childhood cancer survivors
Complete Recovery Care
- Defines “complete recovery care” which includes care to address secondary effects of cancer and its treatment, including late and psychosocial effects
- Coordinates complete recovery care activities across federal agencies
- Establishes a Collaborative that will develop a plan for workforce development for complete recovery care
Section 13: Activities of the Food and Drug Administration
Sense of the Senate
- Encourages the FDA to harmonize policies to facilitate the development of drugs; explore clinical trial endpoints; and, modernize the Office of Oncology Drug Products
____________________________________________
Renewing the War on Cancer
By Edward M. Kennedy and Kay Bailey Hutchison

Kay Bailey Hutchison, U.S. Senator For State of Texas
Cancer is a relentless disease. It doesn’t discriminate between men and women, wealthy or poor, the elderly or the young. In 2008, over 1.4 million Americans were diagnosed with some form of the disease. If it wasn’t you, it may have been a spouse or sibling, a parent or a child, a friend or a coworker. We, too, have known the challenges of cancer diagnoses for ourselves or our family members or friends. And while there are many stories of survival, this disease still takes far too many lives. More than half a million Americans lost their battle with cancer last year.
Since the War on Cancer was declared in 1971, we have amassed a wealth of knowledge about the disease. Advances in basic and clinical research have improved treatments significantly. Some of the most important progress has been made in prevention and early detection, particularly screening, including mammography and colonoscopy. Behavior modifications, such as smoking cessation, better eating habits, regular exercise, and sunscreen have been found to prevent many cancers. Continued focus must be placed on prevention, which will always be the best cure.
Though heightened awareness and prevention should be emphasized, alone they don’t translate into adequate progress for those with cancer. Since 1971, the cancer mortality rate has decreased by only 6 percent. In the same period, by contrast, mortality rates have dramatically declined for heart disease (by 56 percent) and stroke (by 66 percent). Today, cancer is the second leading cause of death in the United States, exceeded only by heart disease. If the current trend continues, the National Cancer Institute predicts that one in every two men and one in every three women will be diagnosed with cancer in their lifetimes, and that cancer will become the leading killer of Americans.
The solution isn’t easy, but there are steps we should take now if we hope to see the diagnosis rate decline substantially and the survival rate increase. To do so, we must identify and remove the numerous barriers that obstruct our progress in cancer research and treatment.
First, it is essential that cancer be diagnosed at an initial, curable stage. One of the most promising breakthroughs is the monitoring of biomarkers, which leave evidence within the body that alerts clinicians to hidden activity indicating that cancer may be developing. Identification of such biomarkers can lead to the earliest possible detection of cancer in patients.
Second, even if we significantly improve early detection, lack of health insurance and other impediments to care will preclude many Americans from undergoing routine screening. With early screening, the disease may be detected at a treatable stage and dramatically increase the rate of survival. Greater outreach is clearly needed to make screening more available to all, and especially to underserved populations.
Third, we must adopt a more coordinated approach to cancer research. Establishing an interconnected network of biorepositories with broadly accessible sources of tissue collection and storage will enable investigators to share information and samples much more effectively. Integrated research will help accelerate the progress of lifesaving research. The search for cures should also be a cooperative goal. The current culture of isolated career research must yield to more cooperative arrangements to expedite breakthroughs. Our national policy should encourage all stakeholders in the War on Cancer to become allies and work in concert toward cures.
Fourth, as our nation’s best and brightest researchers seek new ways to eradicate cancer, we must improve treatment for those who have it today. Raising awareness of clinical trials would result in more patients and their doctors knowing what promising trials are available. Doing so will expand treatment options for patients, and enable researchers to develop better methods for prevention, diagnosis, and therapy. Today, less than five percent of the 10 million adults with cancer in the United States participate in clinical trials. Disincentives by the health insurance market, preventing patients from enrolling in clinical trials, must be eliminated.
Finally, as our knowledge of cancer advances and patients live longer, we need a process that will improve patient survivorship through comprehensive care planning services. There is great value in equipping patients with a treatment plan and summary of their care when they first enter remission, in order to achieve continuity of therapy and preventing costly, duplicative, or unnecessary services.
We have introduced bipartisan legislation to bring about these necessary changes, and we hope to see the bill enacted in the coming weeks and months. These policy initiatives cannot be fully implemented without broad support and sufficient resources, and we are committed to leading this effort to completion.
It’s time to reinvigorate the War on Cancer, and more effective coordination of policy and science is indispensible for rapid progress.

















 U.S. SBRT Liver Metastases Study
U.S. SBRT Liver Metastases Study